- Home >
- CRS & HIPEC >
CRS & HIPEC
In essence, CRS and HIPEC is a recent surgical technique developed for the treatment of Peritoneal surface malignancies.
What are Peritoneal Surface Malignancies?
Peritoneal surface malignancies are tumours that either arise from the peritoneum itself or that spread predominantly to and along the peritoneal surface.
What is the peritoneum?
Peritoneum is a membrane that lines the inner surface of the abdominal cavity and envelopes the intra-abdominal organs and viscera. The peritoneum is made of two parts, the visceral and parietal peritoneum. The visceral peritoneum covers the internal organs and makes up most of the outer layer of the intestinal tract while the parietal peritoneum envelops the inner aspect of the abdominal cavity.
Peritoneal surface malignancies are basically diseases that tend to spread along the peritoneal surface by a phenomenon called as “Redistribution”. The free cancer cells are released in the peritoneal cavity and are carried in the peritoneal fluid. These cells settle down in the areas of stasis and absorption of peritoneal fluid. Usually, there is a latent period during which they are confined to the peritoneal cavity and amenable to CRS and HIPEC. Once the disease starts spreading outside the peritoneal cavity, it is beyond the scope of surgery and CRS and HIPEC is not possible.

These kinds of tumours present a rather tricky therapeutic challenge since the naturally existing blood-peritoneal barrier (a physiological barrier that separates the blood stream and the peritoneal cavity) physically prevents the chemotherapy administered intra-venously from entering the peritoneal cavity in adequate concentrations to eradicate the disease. Further, the disease has a tendency to spread by the phenomenon of “Redistribution” settling down in areas of stasis and where peritoneal fluid has been reabsorbed. Additionally, in patients who are already operated upon, the post-operative adhesions serve as sanctuaries for carcinomatosis (cancerous) cells since chemotherapy cannot reach those areas because of the poor blood-supply post-surgery. The peritoneum itself receives only 2% of the cardiac output (the amount of blood pumped by the heart in 1 minute); this makes intra-venous administration of the chemotherapy a highly inefficient way of delivering chemotherapy for peritoneal cancer.
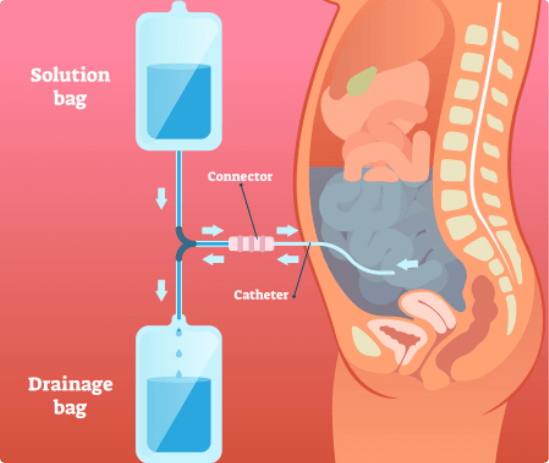
To overcome the above-mentioned challenges, a treatment protocol comprising of a combination of chemotherapy and surgery has been developed in the recent years after several painstaking and systematic experimental and clinical studies.
It takes place in 2 stages.
- Cyto-Reductive Surgery (CRS)
- Hyperthermic Intra-Peritoneal Chemotherapy (HIPEC)
The Cyto-reductive surgery is a “Maximal Effort Surgery” and involves systematic, laborious and thorough physical removal of all visible tumour tissue. This, theoretically, reduces a stage IV disease to an RO (no residual tumor status) status with no macroscopic or visible disease. The microscopic disease that may be left behind after such a long surgery is then further eradicated by the HIPEC (Hyperthermic Intra-peritoneal Chemotherapy). The combination of heat and chemotherapy is proven to increase the intra-cellular concentration of chemotherapy by several times compared to norma-thermic chemotherapy. It is the combination of both these modalities that makes this treatment highly efficient in the treatment of peritoneal surface malignancies.
Treatable Disaeses
With CRS and HIPEC, Peritoneal Surface Malignancies can be treated.
These include:
- Pseudomyxoma peritonei
- Appendiceal cancers
- Diffuse Malignant Peritoneal mesothelioma (DMPM)
- Colorectal cancers with peritoneal metastases (colorectal carcinomatosis)
- Ovarian cancer with peritoneal metastases (especially in the interval setting, recurrent or residual ovarian cancers)
- Primary peritoneal carcinomas
- Gastric cancer with peritoneal metastases or cytology positive gastric cancers
- Select cases of endometrial cancers with peritoneal metastases (cancer of the endometrium of the uterus that has spread to the peritoneum), and other rarer peritoneal surface malignancies like rare ovarian tumours, Desmoplastic small round cell tumors (DSRCT), peritoneal metastases of HCC, granulose cell tumours, etc.

HIPEC Procedures
During the HIPEC procedure, the surgeon will continuously circulate a heated sterile solution – containing a chemotherapeutic agent – throughout the peritoneal cavity, for a maximum of two hours. The HIPEC procedure is designed to attempt to kill any remaining cancer cells. The procedure also improves drug absorption and effect with minimal exposure to the rest of the body. In this way, the normal side effects of chemotherapy can be avoided.
Results
Peritoneal malignancies traditionally have had a poor prognosis with virtually no chance of cure and at best marginal improvement in survival results with palliative chemotherapy.
With CRS and HIPEC, the survival and quality of life has improved dramatically for these diseases. This is a rapidly growing field with the indications expanding rapidly and results that prove beyond doubt the effectiveness of the treatment. For instance,
- For Pseudomyxomas, the traditional treatment results in a 5-year survival of up to 50% at best with a poor quality of life and multiple surgeries. CRS and HIPEC is already established as the treatment of choice with multicentric studies showing a 10-year survival of up to 70%.
- For diffuse Malignant Peritoneal Mesotheliomas, the median survival with traditional treatment strategies is at best 1 year; however, with CRS and HIPEC the median survival in recent series has approached 5 years with more than 50% 5-year survival rates.
- Peritoneal Carcinomatosis of Colorectal cancer origin – the recent French PRODIGE 7 study showed that Oxaliplatin based short duration HIPEC (460 mg for 30 min) is not effective over and above CRS. However, CRS itself gives a median survival of >40 months, which is nearly twice that achieved by just palliative chemotherapy. This is a fantastic outcome and a validation for the role of surgery in peritoneal metastases of colorectal origin. This study did not include mitomycin a HIPEC agent and this needs to be studied further.
- There are several randomised and multi-centric studies that have proven the benefit of CRS and HIPEC for peritoneal metastases of Gastric cancer.
- Similar dramatic results are obtained in recurrent Platinum-sensitive Ovarian Carcinomas, and other rare Peritoneal Malignancies.
- Comprehensive quality of life studies and cost-effectiveness studies have also shown results in favour of CRS+HIPEC.